Edge Cases Aren't Optional, Rare Diseases Aren't Rare
They're just ignored, dismissed, misdiagnosed, psychologized, and sometimes institutionalized
As an amateur coder, I frequently have to anticipate instances in which user behavior might not conform to what is expected. In other words, I have to be prepared for edge cases.
A user might enter non-numerical characters in a phone number field. A form might require conditional logic. A customer in another country might require different payment processing options.
Ignoring edge cases doesn’t make them go away. It just makes you sloppy and bad at your job. You need to be prepared for all the different ways in which users might engage with your site because you don’t get a second chance to make a first impression.
In healthcare, however, it seems perfectly acceptable to shirk this responsibility and deny the existence of edge cases or medical zebras, patients with rare or complex illnesses.
Medicine does not take kindly to zebras, and if you think you’re one, the profession will vehemently insist that you simply need new glasses. You’re just anxious. Stressed. Depressed. Drug-seeking. Attention-seeking. Noncompliant. Argumentative. Difficult.
You get the picture. If a physician can’t figure out what ails you in 20 minutes spread across four outpatient visits, your symptoms are deemed psychosomatic.
Gaslighting and medical PTSD are unfortunately the norm for zebras.
The medical system is perfectly content in catering to the average, which doesn’t really exist in the first place, while minimizing, psychologizing, and stigmatizing the rest.
If you think that dismissal is fringe, you’d be mistaken. It’s as mainstream as white coats in medicine. These attitudes have been pervasive for centuries, not decades.
A Culture of Violence
In 2007, Gerald Weissmann, Professor Emeritus at NYU School of Medicine and then editor-in-chief of the FASEB Journal, wrote an editorial titled “‘Chronic Lyme’ and other medically unexplained syndromes” in which he argued for the psychosomatic etiology of various conditions, including myalgic encephalomyelitis, irritable bowel syndrome, and fibromyalgia.
Curiously, Weissmann drew parallels to female hysteria in a laughable attempt to bolster his position, which could be summarized as, “Remember when female hysteria was a thing? Well, this is the modern-day equivalent, so it’s also psychosomatic.”
Skeptics worry, however, that the hallmark of these “diseases” is that diagnosis requires the complete absence of objective physical or biochemical derangement. They wonder whether such patients are not really victims of a complex set of socially and medically constructed diseases—much as the “railway spine,” “chronic appendicitis,” or “female hysteria” favored by 19th century clinicians.
Female hysteria is a cautionary tale about psychologizing women’s pain, not encountering credulous patients. The term was historically used to describe any number of conditions that primarily affected women, including multiple sclerosis, epilepsy, endometriosis, lupus, and postural orthostatic tachycardia syndrome.
The lesson of female hysteria is not that women imagine illness. It's that medicine has a 200-year track record of ignoring women’s health issues and then gaslighting them about it.
Invoking the notion of female hysteria as a valid argument for the existence of socially constructed diseases is nothing short of misogyny. But I guess when you’re the editor of the journal, you’re allowed to be as historically illiterate and tone-deaf as you like.
Weissman went on to liken chronic fatigue syndrome to Munchausen syndrome by proxy, a form of child abuse and mental illness where a caregiver, often a mother, exaggerates, fabricates, or induces physical or psychological symptoms in a child to gain attention or sympathy.
Chronic fatigue syndromes are also found in children; the condition might be called “Münchhausen’s fatigue by proxy.”
This type of dangerous, uninformed accusation that a child with inexplicable symptoms is simply being abused has resulted in sick children being physically removed from their homes on suspicion of abuse.
Andrew Abrahams and Tracy Finnegan covered this topic in the documentary Complicated, which describes the stories of children with Ehlers-Danlos syndrome, a group of complex, misunderstood conditions affecting everything from bones and joints to blood vessels and the GI tract.
Even when medical documentation and letters from physicians attest to the physical nature of a child’s disease, parents have lost custody of their children based on the opinions of uneducated social workers and doctors.
Weissman erroneously stated that chronic fatigue syndrome and other medically unexplained are more common among children from rich families, which he believed precluded an infectious etiology, as though differential access to healthcare were not an obvious confounding variable.
This is a textbook case of collider bias. Wealthier families tend to have better access to healthcare services that allow for diagnosis, so stratifying on this basis creates a spurious correlation between socioeconomic status and disease. The underlying condition may be more prevalent in poorer populations, but we aren’t measuring it.
Weissman referred to patients with unexplained syndromes as “individuals who enjoy the dignity that all disease confer.” Forgive me, but I fail to see how losing one’s health, home, livelihood, friends and family confers dignity.
Physicians like Weissman feel that patients want to be sick, as though we have nothing better to do than sit around all day being attention-seeking hypochondriacs. As though we don’t have dreams and goals and ambitions of our own. As though we are somehow subhuman.
Not only are these attitudes not rare, but they receive virtually no pushback. Medical culture sanctions violence and violations of autonomy, as Zed Zha, MD (she/her) describes through her writing.
What we condone tells us something about what we value.
Despite his stance on unexplained medical conditions, Weissman was enthusiastically celebrated for his contributions to rheumatology research and lauded as a paragon of medical virtue throughout his life.
If a physician endorsed flat earth theory or denied climate change however, the public response would be outrage and concerns about competency. Tell patients that their symptoms are psychosomatic, and everything is copacetic.
Trial by Error
Since Weissmann viewed patients and advocates as people who “march against medical science,” it’s perhaps no surprise that he invoked none other than British psychiatrist Simon Wessely, psychologizer extraordinaire, creator of the cognitive behavioral model (CBM) of chronic fatigue syndrome, and defender of the abomination that is the PACE trial.
The PACE (Pacing, Graded Activity, and Cognitive Behavior Therapy: A Randomized Evaluation) trial significantly shaped the scientific community’s understanding of ME/CFS, causing irreparable harm to patients by negatively affecting research funding, treatment modalities, and public attitudes towards the illness.
The three principal investigators — Peter White, Trudie Chalder, and Michael Sharpe (two psychiatrists and a psychotherapist) — concluded that adding cognitive behavioral therapy and graded exercise therapy to specialist medical care improved outcomes for people with ME/CFS but adaptive pacing provided no benefit. Although not listed as a byline author, Simon Wessely served as a consultant on the trial.
Many patients and researchers around the world were skeptical of the study results because graded exercise often produced the hallmark symptom of post-exertional malaise (PEM), a delayed exacerbation of symptoms following activity that was previously tolerated.
PEM is distinct from deconditioning, characterized by significant declines in VO₂ max and anaerobic threshold with exertion as measured by 2-day cardiopulmonary exercise test (CPET), the only validated biomarker that currently exists for ME/CFS and also one that notably wasn’t utilized in the trial design.
The lack of 2-day CPET screening allowed for contamination of the patient population, meaning the trial likely enrolled patients with PEM due to ME/CFS as well as patients who experienced fatigue as the result of other medical conditions.
In other words, the trial was testing an intervention on a population it hadn't even bothered to properly characterize and generalizing the results to patients it may never have studied in the first place.
The trial was unblinded, plagued by glaring methodological errors, and failed to produce any objective measurements of improvement among enrolled patients.
Jonathan Edwards, an Emeritus Professor of Connective Tissue Medicine at University College London, called the trial “valueless for one reason: the combination of lack of blinding of treatments and choice of subjective primary endpoint. Neither of these alone need be a fatal design flaw but the combination is.”
He elaborated that “the authors have not been meticulous in trying to avoid bias that might arise. On the contrary they appear to have acted in ways more or less guaranteed to maximize bias.”
After a five-year battle, Alem Matthees, an Australian patient with ME/CFS, succeeded in getting Queen Mary University of London to release the original trial data under the UK Freedom of Information Act.
A re-analysis of the trial data by Matthees and two statistics professors demonstrated that major changes to the thresholds for recovery had inflated recovery estimates four-fold and that recovery rates in the cognitive behavioral therapy and graded exercise therapy groups were not significantly higher than in the group that received specialist medical care alone.
The SF-36 Physical Functioning subscale is a measure that asks participants to rate their limitations while performing daily activities such as running and lifting heavy objects to walking to bathing.
Respondents can answer ‘Yes, limited a lot’ (score 0), ‘Yes, limited a little’ (score 1), or ‘No, not limited at all’ (score 2). The total score across 10 activities is multiplied by five, so the maximum score of 100 indicates no impairment with any type of activity.
To be eligible to join the PACE trial, participants had to score 65 or lower on this scale. The original study protocol defined recovery as a score of 85 or above. The “revised” recovery threshold of 60 was not only far below the original recovery threshold but also beneath the original entry level threshold of 65 for admittance into the trial.
In other words, if patients deteriorated during the course of the trial, they could still be classified as recovered. The revised recovery threshold score was close to the mean score of patients with Class II congestive heart failure.
Despite significant backlash from scientific and patient communities and numerous calls for retraction over the decades, The Lancet defended the study, and the fraudulent findings continued to poison the well of post-infectious disease research.
Clinical Comorbidities or Clusters?
Weissmann endorsed Wessely’s argument that unexplained medical syndromes don’t describe unique clinical entities, that the same cluster of symptoms receives different names depending on which type of specialist sees the patient. What a rheumatologist calls fibromyalgia, a gastroenterologist would call IBS, a neurologist would call chronic fatigue syndrome.
He believed that these unexplained syndromes converged on a psychosomatic origin. But Dr. Lawrence Afrin, a hematologist-oncologist, believes these conditions converge on mast cells. Three years after Weissmann published his editorial, the first set of diagnostic criteria for mast cell activation syndrome (MCAS) appeared in The Journal of Allergy and Clinical Immunology.
Unlike mastocytosis, which is a clonal disorder characterized by overproduction of mast cells by the bone marrow, MCAS is characterized by normal numbers of mast cells that degranulate inappropriately, releasing inflammatory mediators like histamine, heparin, and cytokines. In other words, you don’t have too many mast cells, but the ones you do have are prone to misbehaving.
Because mast cells are present in virtually every tissue and mediate inflammatory responses across every major organ system, MCAS exhibits a high degree of clinical heterogeneity.
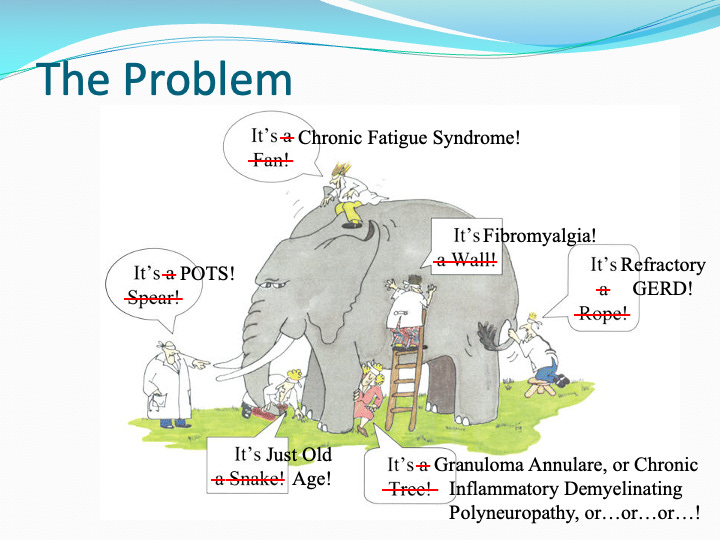
The gastroenterologist might observe irritable bowel syndrome (IBS) or gastroesophageal reflux disease (GERD). Neurology may find small fiber neuropathy (SFN), postural orthostatic tachycardia syndrome (POTS), inappropriate sinus tachycardia (IST), or neurocardiogenic syncome (NCS). The dermatologist notes urticaria and flushing. The allergist finds anaphylactoid reactions without IgE elevation. Each specialist sees a piece of the puzzle. No one sees the bigger picture.
Afrin illustrates this point in many of his presentations using the analogy of the blind men and the elephant, the ancient parable in which each man touches a different part of the animal and names what he feels. In Afrin’s version, each specialist observes a different consequence of mast cell activation syndrome, treating symptoms in isolation while unaware of common drivers.
In 2007, Albert-László Barabási and his colleagues introduced the concept of the human disease network, or diseasome, to address these types of shortcomings. By mapping relationships between diseases based on shared genetic components, they demonstrated that disorders we treat as distinct clinical entities are deeply interconnected at the molecular level.
Barabási elaborated on the implications in an NEJM editorial that same year, coining the term network medicine to describe what a systems-level view of disease could offer that organ-system-based medicine structurally cannot.
Patient-scientist Cortney Gensemer, PhD explains that the clustering of conditions like EDS, POTS, MCAS, and ME/CFS at high rates likely reflects shared biology rather than unfortunate coincidences. Below is a disease network that I created with D3.js to illustrate the concept.
Occam’s razor meets Hickam's dictum. The former espouses the principle of parsimony, insisting that the explanation that makes the fewest assumptions is probably the right one. The latter contends that a patient can have as many diseases as they damn well please.
In practice, neither principle is inherently right or wrong. Depending on a patient’s clinical presentation, one approach or the other might be safer. In cases of complex chronic illness, the tension sometimes dissolves.
The patient with four accumulated diagnoses is experiencing one underlying biological vulnerability expressing itself across multiple systems simultaneously. The multiple diagnoses are Hickam's. The common cause is Occam's.
The diverse clinical presentations of MCAS shouldn’t be taken to imply that every case of POTS or IBS or GERD is due to aberrant mast cell behavior, but if a patient has many relevant diagnoses across multiple organ systems, that should raise suspicion of a potential mast cell-mediated disorder.
It’s also important to avoid the trap of thinking everything is a manifestation of MCAS, as patients with MCAS have also been known to have Hodgkin lymphoma and other serious diseases. If you’re presenting with alarm symptoms, don’t ignore them.
“Medically unexplained syndrome” is not a diagnosis but a placeholder, a reason to keep looking. Weissmann treated uncertainty as a reason to psychologize, stating that afflicted patients are “in very real pain— but of the mind—and the mind chooses symptoms that will be taken as evidence of physical disease.”
A model that only recognizes inputs it was designed for isn't intelligent but overfitted. Medicine built its diagnostic frameworks around the most measurable manifestations of disease, called everything beyond that range unexplained, and then gaslit patients unfortunate enough to find themselves on the outside.
The edge cases didn't go away. They filled waiting rooms, filed for disability, and turned to online patient forums to compare notes with other outcasts. It’s high time that we mind the gap and expand our range.